- Home
- Details
- Registry
- RSVP
- Where does affinity photo download the samples
- Java for mac 10-7-5 download
- Download and install microsoft word 2013 free
- Songs with magic theme
- Hp dv6000 nvidia drivers for windows 7
- Delete section breaks in word 2016
- Justin timberlake new album zip download
- Windows xp boot disk usb rufus
- Hp smartaudio conexant audio device could not be found
- Adobe creative suite free
- Rainbow six siege pc download
- Install ringcentral app
- Raxco perfectdisk official website
- Rustom song
- How to create a folder on mac for videos
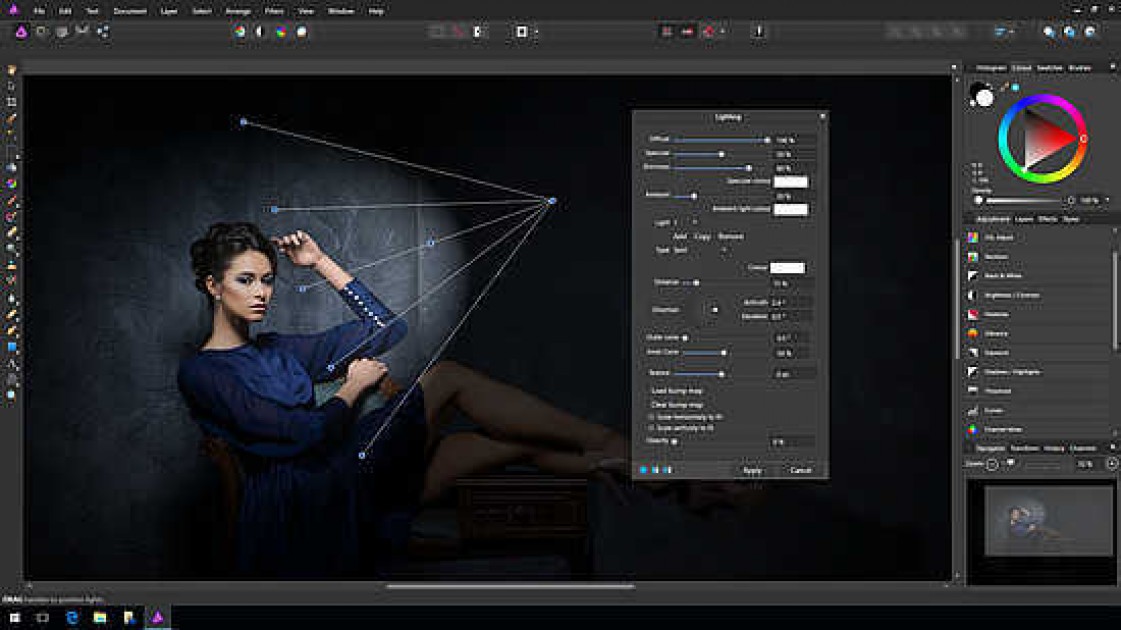
Some of the methods are similar to affinity chromatography by use of immobilized ligands.Īn example of an agarose gel after electrophoresisĪ type of electrophoretic mobility shift assay (AMSA), agarose gel electrophoresis is used to separate protein-bound amino acid complexes from free amino acids. For enzymes and other ligand-binding proteins, one-dimensional electrophoresis similar to counter electrophoresis or to "rocket immunoelectrophoresis", affinity electrophoresis may be used as an alternative quantification of the protein. The methods have been used for estimation of binding constants, as for instance in lectin affinity electrophoresis or characterization of molecules with specific features like glycan content or ligand binding. Nucleic acids or nucleic acid fragments may be characterized by their affinity to other molecules. Membrane proteins may be identified by a shift in mobility induced by a charged detergent. The interaction or binding of a molecule, charged or uncharged, will normally change the electrophoretic properties of a molecule. The methods are based on changes in the electrophoretic pattern of molecules (mainly macromolecules) through biospecific interaction or complex formation. The methods include the so-called electrophoretic mobility shift assay, charge shift electrophoresis and affinity capillary electrophoresis. Both qualitative and quantitative information may be obtained through affinity electrophoresis.
The analysis was performed early in the 1970s at the Protein LaboratoryĪffinity electrophoresis is a general name for many analytical methods used in biochemistry and biotechnology. Electrophoresis performed overnight at less than 10 V/cm. The quantitative principle of affinity electrophoresis illustrated with electrophoresis at pH 8.6 of concanavalin A into an agarose gel containing blood serum (3.6 microliter per square cm).
- Home
- Details
- Registry
- RSVP
- Where does affinity photo download the samples
- Java for mac 10-7-5 download
- Download and install microsoft word 2013 free
- Songs with magic theme
- Hp dv6000 nvidia drivers for windows 7
- Delete section breaks in word 2016
- Justin timberlake new album zip download
- Windows xp boot disk usb rufus
- Hp smartaudio conexant audio device could not be found
- Adobe creative suite free
- Rainbow six siege pc download
- Install ringcentral app
- Raxco perfectdisk official website
- Rustom song
- How to create a folder on mac for videos
